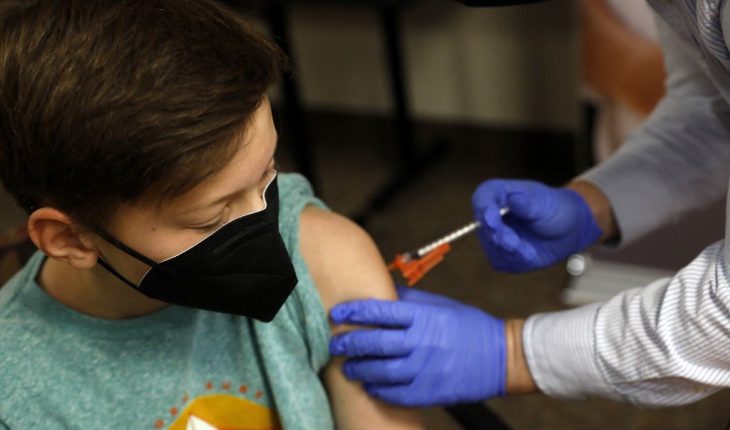
The United States plans to begin vaccinating children aged 5 to 11 against COVID-19 starting in November, the Joe Biden administration said Wednesday, a move that will make 28 million more people immunized in the country.
“Our planning efforts mean we will be prepared to begin applying vaccines in the days following a final recommendation from the Centers for Disease Control and Prevention (CDC),” the White House said, alluding to the country’s top federal public health agency.
Read: Six states begin vaccinating minors with comorbidities
Drugmaker Pfizer applied for permission to apply its covid vaccine in this age group earlier this month, and the issue will be debated by two panels of experts.
The first, convened by the U.S. drug regulator FDA, will meet next week, and the second, convened by the CDC, will meet on Nov. 2 and 3, with the agency expected to make its recommendation soon after.
During a clinical trial, children aged 5 to 11 years received two doses of 10 micrograms, compared with 30 micrograms for older age groups. The injections were given 21 days apart.
The vaccine developed by the Pfizer/BioNTech alliance received full FDA approval for people 16 and older, and the FDA in May authorized its emergency use in children ages 12 to 15.
Experts say vaccinating children is essential to help achieve the population’s immunity against the disease.
While younger children are less likely to develop severe cases of COVID-19, they can still get sick and transmit the virus to the rest of the population.
Confidence in vaccines in the United States has increased in recent months.
As of Wednesday, 77.1% of the currently eligible population age 12 and older had received one or more doses of the covid vaccine.
What we do at Animal Político requires professional journalists, teamwork, dialogue with readers and something very important: independence. You can help us keep going. Be part of the team.
Subscribe to Animal Político, receive benefits and support free journalism.#YoSoyAnimal